Computer Assisted Implant Surgery is increasingly popular, with almost every implant system racing to deliver the most reliable technology. Either as static or dynamic, CAIS promises unprecedented accuracy in implant placement and evidence now shows that it delivers. But is it just about accuracy? Is it worth the cost and trouble? Can this technology transform the way we practice implant dentistry,or will it remain a premium gadget mainly targeting
complex cases in ‘up-market’ clinics?
If you share these questions, then the article that follows is for you!
By Nikos Mattheos, DDS, MASc, PhD
Atiphan Pimkhaokham, DDS, OMFS Specialist, PhD
James Chow, BDS, MDS, OMFS Specialist, FRCDS(Eng), FRCDS(can)
What a difference a mm makes?
Accurate implant placement was the primary and most important advantage reported for CAIS. To measure the extent of this accuracy, researchers have used the 3-dimensional deviation from the planned position at the implant platform and apex and the deviation in the angle (Figure 1). Initial research pointed out to a significant increase in accuracy when CAIS was used, with the deviation reduced to roughly half of what it was in freehand implant placement for single implants. An earlier systematic review from the ITI Consensus Workshop of 2017, found that static CAIS would result to deviation of 1.2 mm at implant platform on average (1), suggesting that a safety margin of 2mm is to be considered when we use this technology. Although certainly a great development, is this something that could transform your practice? Many colleagues remained skeptical, especially those with long experience. After all, gadgets are always cool, but we’ve been placing implants with great success already. Is it worth all additional investment and effort?
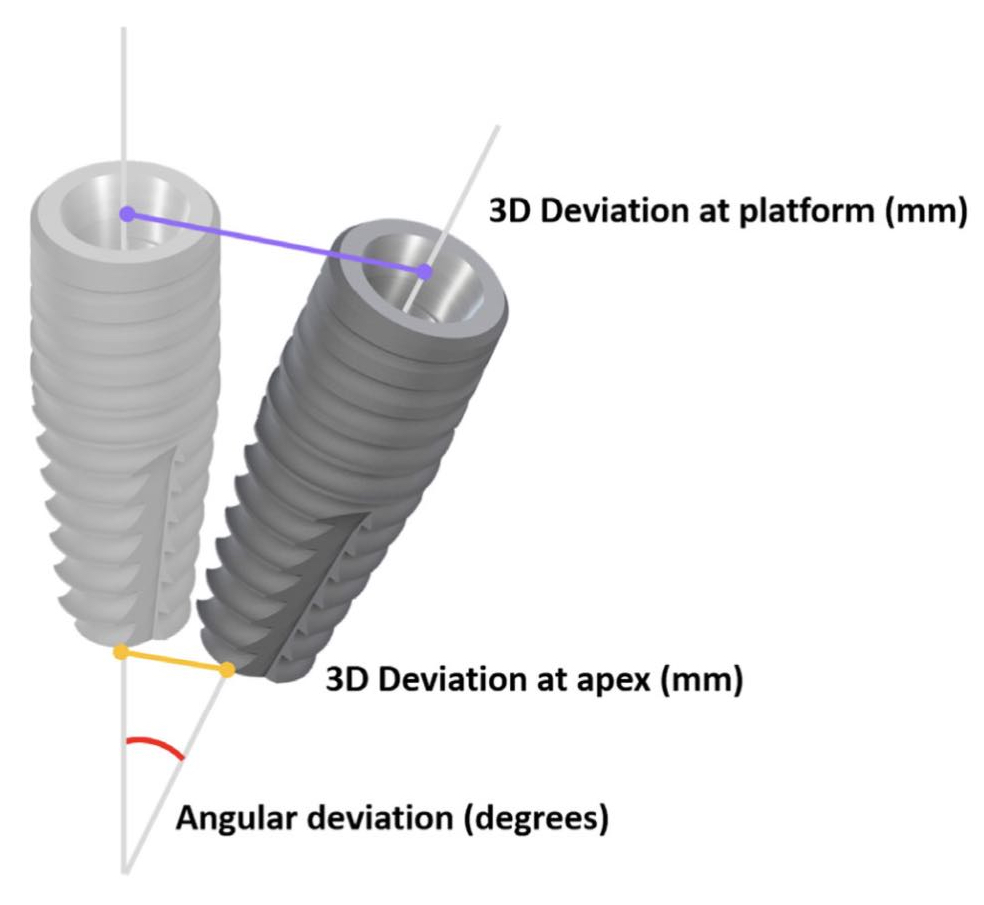
Assessing implant placement accuracy
Typical parametres measured to assess accuracy of implant placement: 3D deviation at platform and apex and angular deviation.
Shifting Paradigms – shifting needs
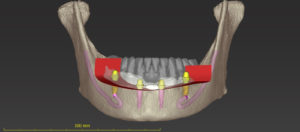
Before we start discussing about accuracy there is one important question each should ask oneself: “how critical is to achieve precisely my planned position?” The answer might be different for each patient case and was different through the different ages in the evolution of implant dentistry. In the earlier decades of Implant Dentistry, when the dominant paradigm was the seek for osseointegration, the placement was driven by the anatomy. Exposing an edentulous jaw, the surgeon would determine the final position based on his assessment of the sites that could offer best conditions for osseointegration. Then, several months later someone would come and join all the implants – wherever placed – under one full-arch reconstruction. As you can understand, reconstructions varied significantly, the plan was only approximate – at least by today’s standards – deviations of implant position could extend to centimetres, while even the number of implants could change intraoperatively. Once replacement of single units became established, the margin of acceptable deviation was narrowed, and the implant placement became increasingly driven by the prosthetic needs. The development however of more sophisticated prosthodontics, (abutment dimensions and angles, retention methods) allowed still a significant “leeway” for implant positioning. Today, implant dentistry has completely departed from the older paradigm. Modern surfaces and implant design, as well as bone regeneration techniques have made osteointegration to be no longer the key determinant. The guiding paradigm today in implant dentistry is this of designing a new organ, which will serve function and aesthetics but also where mechanical components, human tissue and bacteria can reach a long term balance of health (2). This requires a lot of attention to be given to the proper design of the prosthesis, the shape and morphology of the Implant Supracrestal Complex and finally defining the single optimal implant position and type. Even more, the rising concept of immediacy, with an immediate temporary or loaded prosthesis adds critical importance to the implant position. Even a small deviation from the planned position can be detrimental to immediate loading when a pre-fabricated prosthesis is used. So ask yourself, how important is it to reach the planned position with high accuracy? If you perceive the implant-prosthesis complex as one system in close interrelation with the tissue, the answer is that implant position is always critical. Today, we always need to have defined the optimal implant position. The tolerance for deviation and consequently the need for accuracy might differ, depending on the site and the treatment plan. In the case of a single posterior implant to be restored conventionally, we might be able to get away with some significant deviation through “damage-control” prosthodontics. On the other extreme, in an anterior aesthetic case or a full arch planned for immediate restoration, even a small deviation would jeopardise the whole plan. And yes, 1 mm can be the game changer indeed in many cases.
... where half a milimetre maters..!
CAIS Technology
The CAIS tree started initially with two main approaches, the “static” or “guided” and the “dynamic” or “real time navigation”. As with everything new, the terminology is a bit confusing at times, in particular in studies comparing CAIS with conventional placement. So in some studies you will see the conventional placement to be reported as “freehand”, but that will be misleading as the placement with real time navigation is also “freehand”. In other studies the conventional placement was referred to as “mental” guided, but that is misleading too, as mental capacity should be behind any kind of placement. On top of that, with the meaning of “mental” in colloquial American English and with the mental pressure we have all been subjected to under the years of Covid-19, this term is not particularly attractive. So, to streamline our terms a bit, CAIS implies the use of digital aids or products during the surgical implant placement. It can be static, where the surgeon places the implant in a pre-determined position by means of a surgical guide, or it can be dynamic, (which is actually freehand), where the surgeon places the implant by means of real time projection of the osteotomy on a screen, in relation to a pre-identified position. These two technological principles have now evolved and diversified so much, that we can describe a whole “technology tree” with many branches already.
The static CAIS technology
The main idea behind the static CAIS is rather simple: create an acrylic “template”, which will guide our drills to the perfect osteotomy and then place the implant accordingly. It is based therefore on a 3D printed surgical guide with “sleeves”, which allow the placement of the drills controlling angle and position and “stops” to control the depth. The guide can be supported on teeth, on oral mucosa, on the underlying bone through mini screws or a combination of the previous. It can be a single piece guide, or “stackable” in order to cover several procedures, such as for example a possible ostectomy prior to implant placement or a lateral sinus floor augmentation. But this is about where the principle ends and the application starts! Today almost every major implant brand has its own guided surgery software, instruments and overall protocol (Figure 2). The differences can be significant and this diversity is not based on evidence, but rather reflects each company’s philosophy, design, historical features and insider-view on implant surgery. Does it really matter? Well, evidence shows that it does. In a recent study we compared the precision of implant placement with 5 different commonly available guide designs and guided surgery configurations (3). The results showed significant differences, with the “sleeve in sleeve” guides achieving almost half the deviation than some of the “mounted” or “integrated” sleeve guides (Figure 2). At the same time, accuracy might be not the only parametre that is clinically important in a system. Ease of use by the surgeon, reliability of the full workflow, availability of a wide array of drills, sizes and components, durability and costs are also to be accounted for. As we begin to understand the implication of design parametres through proper research, guided surgery kits in the future might start to converge towards the best practices or principles which deliver the most. For now however, be mindful, there is not one such think as “static guided surgery protocol”, despite what some systematic reviews will try to convince you. On the contrary, there are many different protocols, with significant differences in results, potential and limitations. Choose wisely!
The dynamic CAIS technology
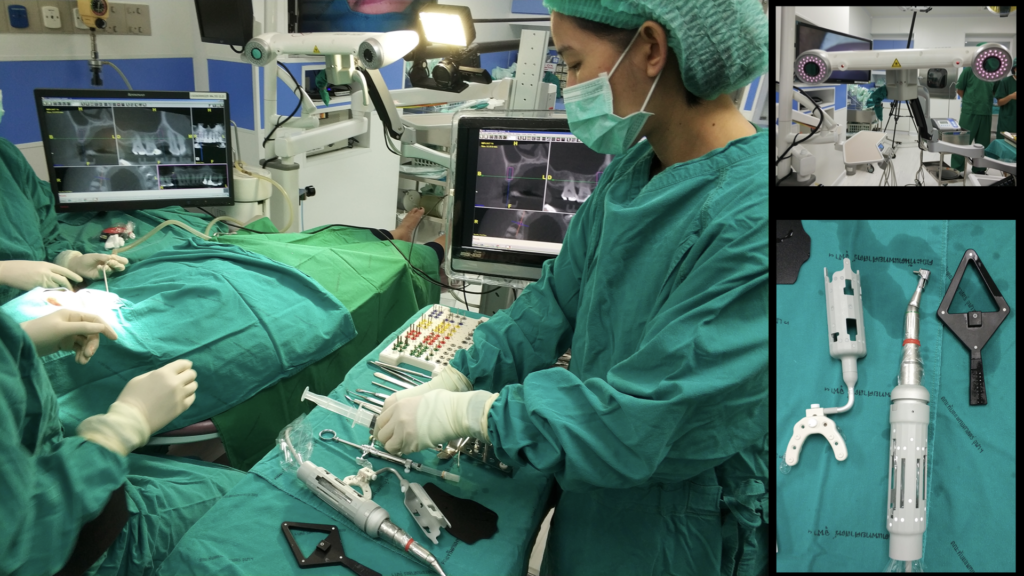
The dynamic CAIS systems are based on a principle which reminds the GPS navigation. In the first generation systems, a set of stereoscopic cameras was constantly recording the position of a set of trackers (called fiducial markers) on the patient and the surgeons handpiece. The software then calculated the exact location of the patient and the handpiece in space and projected the drills on the actual patient’s CBCT in real time, through a screen (Figure 3). Of course, this only made sense if the software could maintain at all times a proper spatial alignment of the patient and the drills. To achieve this, the patient had to wear a custom-made split during the CBCT and during surgery, but also sensitive registration and calibration procedures were required before the surgery. The first and second generation of dynamic systems utilised many components and required rather big areas of fiducial markers (Figure 3). Like in static CAIS also here, there is not a uniform technology and many companies have developed different systems. In this way, there has been systems with 2, 3 or 4 tracking cameras, different size and design of fiducial markers and calibration procedures, as well as different planning and visualization software. There is very little reported in research with regards to comparisons on different dynamic CAIS systems, so one can only try to assess systems performance based on their reported specifications.
The third generation of navigation systems has reduced the need of an intraoperative splint for the patient and instead aligns the surgical visualization via an algorithm directly with the CBCT, using distinct anatomical landmarks on teeth. In general, the trend has been to reduce the number of components as well as streamline the design to smaller and easier to use. One of the latest developments is the addition of augmented reality glasses, which has made the need for chairside operating monitor redundant. This is a major improvement for both speed and ergonomics, as the surgeon has now the navigation screen projected in the upper segment of his view and can perform the surgery without interrupting optical contact with the surgical field. Combining the augmented reality glasses however with surgical loops and light might be a challenge. Learning to operate with all these gadgets mounted and a 3-layer field of vision would require some learning and adaptation (Figure 4).
Precision and Accuracy
As the CAIS systems were initially produced to help clinicians transfer the planned implant position in the mouth, it is not surprising that the first objective of research was to test them for Precision and Accuracy. In daily communication these two terms are often used interchangeably, but when it comes to scientific use, they imply two different qualities (Figure 5). Accuracy will assess how close your placement will be to your planned optimal position. Precision on the other hand will assess how reproducible is the process, that means if you repeat the same placement many times, how close will each placement be to the previous ones.
In practice, they are both important for a CAIS system but for different purpose. A system of high accuracy is a system that fits the purpose and will ensure the implant is placed in the planned position. A system of high precision on the other hand will be a system which is streamlined and not vulnerable to any “systemic” or random influences. These influences could be anything, from calibration of the software to preferences of the operator (right or left handed), or accessibility for instruments.
A good system will need to be checked for both. A more common condition, low precision but high accuracy will reveal some systemic error, for example some error in the software, imprecise calibration or some mistake of the operator.
To test accuracy, we would typically rely on clinical studies, which are the most relevant. Many studies are available on accuracy and we presented some in a previous discussion. However, in clinical studies we can only place each implant once, so to assess precision we would need simulation studies, where the implant is placed in multiple identical models under the same conditions and by the same operator. Try this for your own system to see how precise it gets.
Before we estimate the accuracy of CAIS however, we will need to note that the actual guided surgery is only one final step of a long sequence in a digital workflow. This workflow includes a lot of steps, each of them with potential for errors. First, we rely on a CBCT. Unlike what you might think, the 3D image of the CBCT software we work with in our computer screens is not a radiographic image. It is an algorithmic reconstruction of many radiographic images. A periapical radiograph taken with the parallel cone is a true radiograph, as the beam of rays is what actually “writes” on the radiosensitive plate. Any distortion in size is directed by the laws of physics. In the case of a CBCT however, a large number of radiographs is “sutured” together in a seemingly 3D structure by a mathematical algorithm. Every algorithm will use at certain points some “imagination” to fill in the gaps between the radiographs, a process which is a calculated “approximation” and might not always be absolutely loyal to the true anatomy. Adding to that some well-known limitations due to position of the beam, rotation, distances from the source of the rays and more, one can easily understand that although a CBCT is a highly accurate reconstruction for measurements, there is an inevitable error which might differ from machine to machine, day to day and even patient to patient. Some studies have found that linear measurements on well calibrated CBCT can have as much as 0.6mm inaccuracy from the true anatomy (4), while volumetric measurements can deviate as much as 4.4% (5).
Importing the DICOM file to the treatment planning software invites one more algorithm in the party. Then transposing the STL file from the optical scan and the essential rendering has the potential to further influence dimensions. Finally, 3D printing of a surgical guide is a workflow step with potential to introduce some more distortion.
Conclusively, by the time we are about to use our CAIS system to place the implant, there is already some accumulated error from the workflow, that even if the CAIS was to be perfect, we would still observe some deviation from the planned position. How much of the deviation is attributed to the actual CAIS and how much to each of the previous workflow steps is next to impossible to find, especially considering that each software algorithm is a “black box” for us clinicians and researchers. We just have to bear in mind that every time we compare two CAIS systems, we do not compare just two drilling protocols. We essentially compare two complete workflows, including the respective software and all algorithms used. At the same time, it becomes apparent that comparing accuracy estimates between different CAIS systems is meaningful when the workflow is identical, which includes the CBCT and planning software, as well as the operators and clinical setup.
Accuracy of CAIS systems
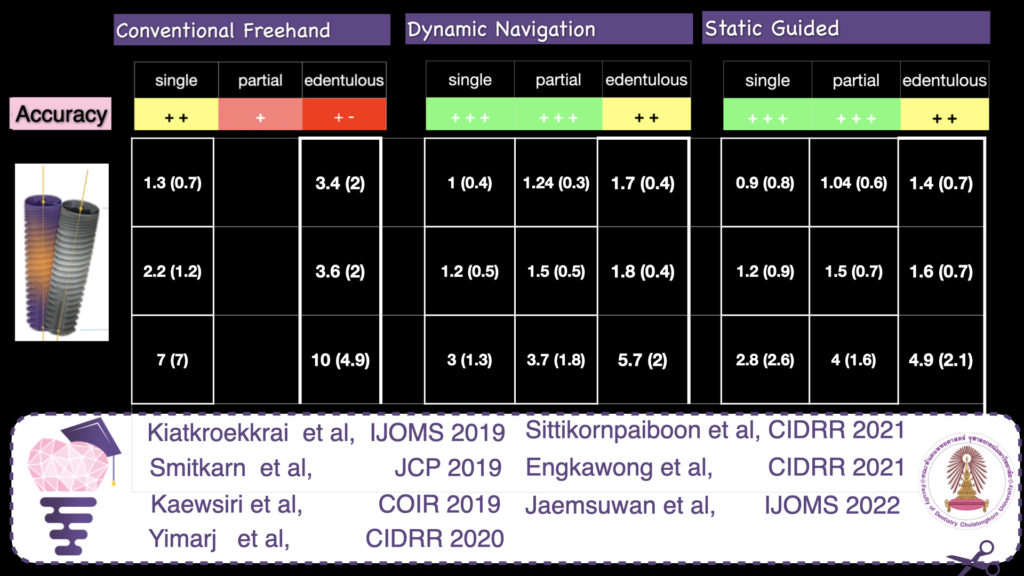
Back the main point now: how accurate is implant placement when we use CAIS? To answer this allow me to discuss the results we have accumulated through a major CAIS research project in our clinic at Chulalongkorn University in the past 3 years (3,6-11). This project has by now involved more than 200 patients and included research protocols in many different clinical scenaria. The strength of this project is that it allows us to shape a comprehensive view of the effectiveness of CAIS, while limiting as much as possible the variation of the workflow error. A typical systematic review will compile numbers from many published studies, e.g. 2 studies from centre A on single gaps and 1 study from centres B and C each on fully edentulous patients. It would be very difficult to draw any valid conclusions on the accuracy of CAIS in single gaps as opposed to fully edentulous, because the 3 studies essentially report the results of 3 different workflows. The three centres use a different CBCT machine, a different planning software and in the case of static CAIS they might be using different printers and guided surgery implant systems. With all these different parametres contributing differently to the potential errors, it is impossible to conclude what is the impact of the clinical condition (single gap vs fully edentulous) in the finally reported accuracy numbers.
By standardizing the studies however and using the same technology and software, the same operators , clinic set up, as well as the same patient pool like we have done in this project, we can at least ensure that the workflow error is similar in all cases thus the observed differences can be attributed to the clinical conditions we investigate.
And at last, time to talk some numbers, which you can see in Figure 6. The table is compiled out of a series of published studies in our centre, where all cases were treated by the same team, using the same CBCT machine as well as planning software and implant system. Streamlining the workflow, one can now see a clear trend with the accuracy of CAIS being closer to freehand at single gaps, while the deviation drastically increases as we move to partially and fully edentulous patients. The deviation also trends to be slightly higher in dynamic than static guided when we are dealing with partially and fully edentulous cases, but at the same time both static and dynamic CAIS perform increasingly (and significantly) better than freehand placement, as complexity increases.
With regards to precision now, one would have to utilize simulation studies analysing the distribution of the deviations in repeated implant placement using the same workflow as we used in the clinical studies above.
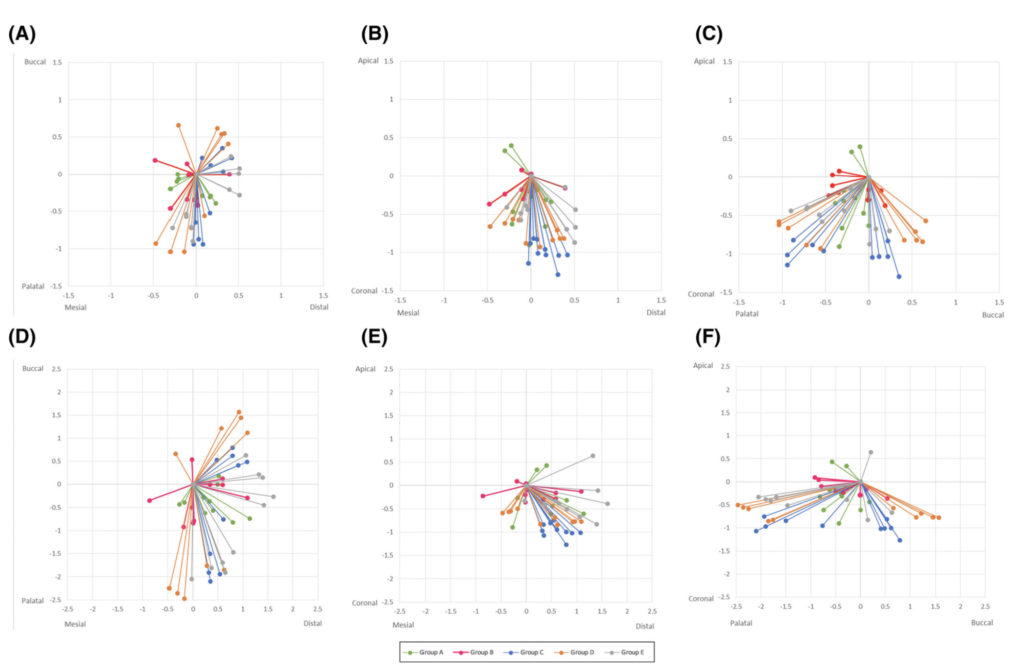
In a simulation study with multiple placements, the deviations of each placement ideally should be scattered equally around the optimal centre, like a symmetric firework. In reality however deviations tend to cluster more towards some sides, pointing to some systemic influence mostly attributed to the operator such as right- or left-handed, ergonomics, optical field as well as specific anatomic locations of the implant site and more. How much clustering of the deviation is acceptable and when should we seek and correct the systemic influence? Well, this is a wider discussion and it should be always be seen in conjunction with the accuracy readings. Maybe we can discuss this in more detail in the future.
The image below is from one of our simulation studies and it shows the clustering of the deviations (precision) for the standard level of accuracy. As you can see, although not too bad, our firework looks like it was fired on a very windy day in some instances…! To understand more why this happens however, you will have to read the paper!
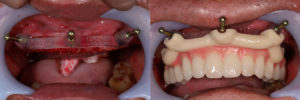
Nevertheless, although precision and accuracy remain the first and primary research focus, they are just the “tip of the Iceberg” when it comes to utilizing CAIS. Other clinical outcomes are also very important such as implant stability, complication rates, invasiveness. Decision making also relies on the overall efficiency of each method, cost/effectiveness, time allocation, initial investment and not to forget the importance of the patients’ own experience, understanding and perceptions. The picture is also completed by anatomic conditions and the treatment objectives, which might also play a critical role in determining the technology that works best in each case. Finally, combining all these factors we can produce a decision-making tree helping us to assess the individual patient and select the most appropriate CAIS protocol where it offers the greatest advantage.
…but let’s make a pause here. Decisions need also to account for anatomic conditions and the treatment objectives, which will play a critical role in determining the technology that works best in each case. If you are interested to hear more on all these, we will have to return with a second article in the future!
If you want to learn more about computer assisted implant surgery and stay in touch, leave a comment below or drop your email in the form!
References
1. Ali Tahmaseb, Viviane Wu, Daniel Wismeijer, Wim Coucke, Christopher Evans. The accuracy of static computer-aided implant surgery: A systematic review and meta-analysis
Clin Oral Implants Res. 2018 Oct;29 Suppl 16:416-435.
2. Mattheos N, Vergoullis I, Janda M, Miseli A.
The Implant Supracrestal Complex and Its Significance for Long-Term Successful Clinical Outcomes. Int J Prosthodont. 2021 Jan-Feb;34(1):88-100. doi: 10.11607/ijp.7201.
3. P. Sittikornpaiboon, S. Arunjaroensuk, B. Kaboosaya, K. Subbalekha, N. Mattheos, A. Pimkhaokham.
Comparison of the accuracy of implant placement using different drilling system for static Computer-Assisted Implant Surgery: a simulation-based experimental study
Clin Implant Dent Relat Res. in press 2022
4. Pimentel PA, Bomfim RT, Andrade LC, Ferraz EG, Ribeiro Lamberti PL, Rubira-Bullen IR, Sarmento VA.Dimensional error of three-dimensional images generated by different software.
J Med Eng Technol. 2015;39(8):480-4
5. Park CW, Kim JH, Seo YK, Lee SR, Kang JH, Oh SH, Kim GT, Choi YS, Hwang EH.
Volumetric accuracy of cone-beam computed tomography. Imaging Sci Dent. 2017 Sep;47(3):165-174.
6. S Engkawong, N Mattheos, P Pantumwadee Pisarnturakit, A Pimkhaokham, K Subbalekha. Comparing patient-reported outcomes and experiences among static, dynamic computer-aided, and conventional freehand dental implant placement: A randomized clinical trial.
Clin Implant Dent Relat Res. in press 2021
7. Yimarj P, Subbalekha K, Dhanesuan K, Siriwatana K, Mattheos N, Pimkhaokham A.
Comparison of the accuracy of implant position for two-implants supported fixed dental prosthesis using static and dynamic computer-assisted implant surgery: A randomized controlled clinical trial.Clin Implant Dent Relat Res. 2020 Sep 17. doi: 10.1111/cid.12949.
8. Kiatkroekkrai P, Takolpuckdee C, Subbalekha K, Mattheos N, Pimkhaokham A.
Accuracy of implant position when placed using static computer-assisted implant surgical guides manufactured with two different optical scanning techniques: a randomized clinical trial.
Int J Oral Maxillofac Surg. 2019 Sep 19.
9. Smitkarn P, Subbalekha K, Mattheos N, Pimkhaokham A.
The accuracy of single-tooth implants placed using fully digital-guided surgery and freehand implant surgery. J Clin Periodontol. 2019 Sep;46(9):949-957
10. Kaewsiri D, Panmekiate S, Subbalekha K, Mattheos N, Pimkhaokham A.
The accuracy of static vs. dynamic computer-assisted implant surgery in single tooth space: A randomized controlled trial. Clin Oral Implants Res. 2019 Jun;30(6):505-514.
11. Jaemsuwan S, et al.
Comparison of the accuracy of the implant position among freehand, static and dynamic computer assisted implant surgery in fully edentulous patients: an experimental study.
IJOMS, under review.








5 thoughts on “Computer Assisted Implant Surgery in Clinical Practice!”
Great summary article on the state of present Guided surgery.
Seems like dynamic guidance has a lot of potential and should allow more real time adjustments if needed.
At the same time, we would like young dentists to learn how to plan because any guidance only takes you to where you plan to place the implant in the first place.
Thank you for your comment! Indeed, I also think the dynamic has great potential as technology becomes smaller and cheaper…actually I recently saw an application of this technology (stereoscopic cameras+ software) for the registration of dynamic jaw movements..!
Have any membership options?
Thank you for your interest! Please leave your email and we will keep you updated of any upcoming articles and news!
Thank you for giving a clear summary of the difference in computer guided surgery. Please keep me updated on any future results on accuracy of the Dynamic Guided surgery technique